
If two light nuclei merge, they can form one heavier nucleus and release energy in the process. The example below shows how another nuclear equation can be written. The atomic numbers and mass numbers on either side of this equation balance, which means that the fission can indeed occur in this manner. The reactants are on the left side of this arrow and the products are on the right. Note that this equation contains a rightward-facing arrow rather than the equality symbol. The mass and atomic numbers on both sides of a nuclear equation must balance for the nuclear reaction to occur. This is called a nuclear equation and the equation for the example described looks as follows. Now, if we consider the previous example of the splitting of uranium-235 into barium-139 and krypton-95, we can write an equation for the reaction to represent the balance of the reactants and products. Where the atomic number Zrepresents the number of protons in the nucleus of this atom and the mass number Arepresents the number of protons and neutrons in the nucleus. We must first recall that the symbol for any neutral atom Xis written in the following way: The process continues in an uncontrolled chain reaction, Wikimedia Commons CC BY-SA 4.0 Nuclear fission example The two neutrons produced are then used to cause the fission of two further uranium-236 nuclei. Uranium-236 undergoes fission to produce barium-144 and krypton-89. The fission diagram below shows the uncontrolled chain reaction that would occur from the fission of uranium-236. This is how nuclear fission reactors are created using uranium. If only one product neutron is used to cause the fission of one nucleus, then the chain reaction is controlled. If we could control the number of product neutrons used to initiate further fissions then we could control the total amount of energy that is released. Just a small amount of uranium undergoing fission has the potential to destroy an entire city. It is this principle that was used to build the atomic bomb. This is called an uncontrolled chain reaction and can release an enormous amount of energy in a short amount of time. The number of fissions will therefore grow exponentially with time. These two neutrons can cause two further uranium nuclei to undergo fission producing four neutrons which may cause four fissions to occur. The example above showed uranium-235 undergoing fission to form barium-139 and krypton-95 and two neutrons. This process is called a nuclear chain reaction. These neutrons that are produced may then be used to cause further nuclear fissions. Moving neutrons are responsible for fission to occur and the fission products include neutrons with kinetic energy. The example below shows how fission occurs. Part of the energy that is released is in the form of the kinetic energy of the fission products. The smaller nuclei, known as the fission products, are usually also unstable and may release alpha or beta particles to attain stability. The nucleus then splits into two smaller nuclei, that are similar in size, and releases two or three neutrons in the process along with large amounts of energy in the form of gamma rays. After the neutron collides with the nucleus, it causes the nucleus to become unstable. It requires a neutron to collide with the heavy nucleus for the splitting to occur. This process is known as nuclear fission and is not random. Large nuclei can also become more stable by splitting into two smaller nuclei. This process occurs spontaneously and randomly. Some heavy atoms are unstable and they maintain stability by emitting radioactive particles such as alpha and beta particles. Heavier nuclei can also be split in a process called nuclear fission, which produces smaller nuclei and some energy. This process is known as nuclear fusion, during which, energy is released. Smaller atoms can sometimes combine to form heavier atoms when their nuclei collide and merge. 
In this article, we will discuss the splitting and combining of nuclei, also known as fission and fusion respectively. Since humans have learned that nuclei can be split apart or fused, we have been curious to understand the energies involved in these reactions. These tiny components are responsible for some of the most energetic (and destructive) reactions that occur on Earth. Heavier atoms consist of heavier nuclei, that is, there are more protons and neutrons. 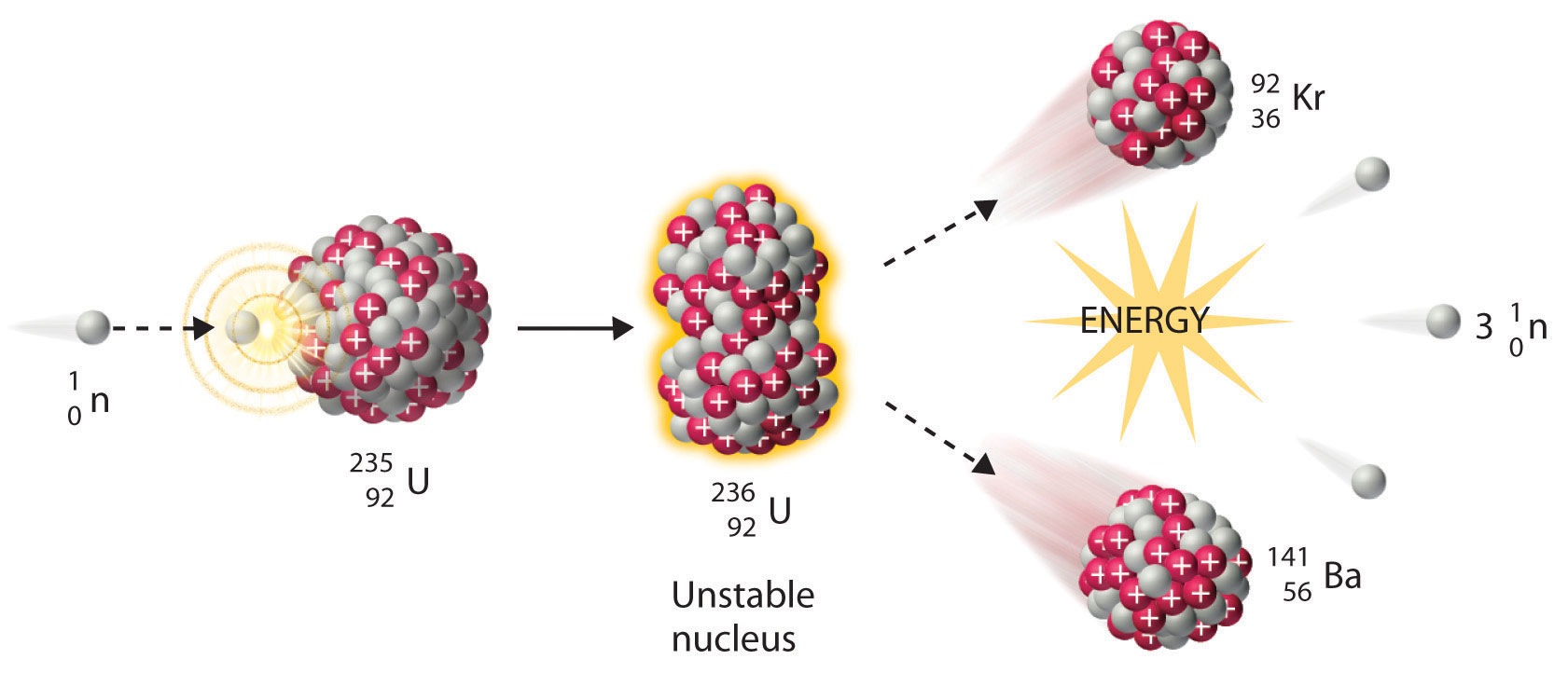 
It consists of neutrons and protons that are held together by a strong nuclear force. This is the core of the atom and constitutes more than 99% of the atom's mass. At the centre of every atom lies the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
